NIH and the biomedical research enterprise have a long history of international collaborations with rules of engagement that allow science to advance while assuring honesty, transparency, integrity, fair merit-based competition, and protection of intellectual capital and proprietary information. These rules of engagement also are designed to limit bias in the design, conduct, and reporting of NIH-supported research. This page describes actions that NIH, institutions, and researchers can take to protect against inappropriate foreign interference. The principles described here align with those announced by the White House Office and Science and Technology Policy in June 2020 (PDF) and in the NSPM-33 implementation guidance.
Overview
NIH research is built on a set of bedrock principles of scientific excellence, unassailable integrity, and fair competition. NIH expects applicants for and recipients of NIH-supported research — both domestic and foreign — to abide by these principles.
It is critical for NIH-supported institutions and their researchers to be wholly transparent about financial support from and affiliations with international institutions. Such transparency ensures that NIH's funding decisions are fair and appropriate, and that U.S. institutions and the American public benefit from their investment in biomedical research.
The biomedical research workforce continues to be greatly enriched and strengthened by scientists working together from many parts of the world. The extraordinary contributions of foreign nationals to American science are indisputable. The overwhelming majority of researchers participating in NIH grants, both in the U.S. and in other countries, are honest contributors to the advancement of knowledge that benefits us all. The research community must find ways to build and continue these important and successful relationships with foreign scientists around the world while simultaneously protecting the Nation's biomedical innovations and intellectual property.

About Foreign Interference
Learn about U.S. government concerns related to inappropriate interference by foreign governments and institutions over federally funded research.

Requirements for Disclosure of Other Support, Foreign Components, and Conflicts of Interest
Learn about applicant and recipient institution responsibilities as well as which activities need to be disclosed and where and when to disclose them.

Format Pages, Instructions, and Samples
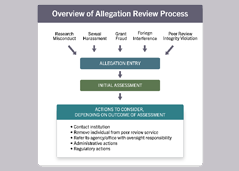
Process for Handling Allegations
Learn about NIH's standard procedures for handling allegations related to inappropriate foreign interference.

Notices, Statements and Reports
Find related notices, policy references, statements, reports and other resources from NIH, the Office of Science and Technology Policy, the Department of Justice, and others.

Case Studies
Read case studies based on real experiences related to foreign interference activities.

Data
NIH provides semi-annual updates on foreign interference cases.