Notice Number: NOT-OD-06-064
Key Dates
Release Date: May 3, 2006
Issued by
National Institutes of Health (NIH), Office of Extramural Research (http://grants.nih.gov/grants/)
The Office of Extramural Research, NIH, is seeking comments regarding possible revisions to certain fiscal policies that govern the Ruth L. Kirschstein National Research Service Award (NRSA) programs, which comprise institutional research training grants (T32, T34, and T35) and individual fellowships (F30, F31, F32, and F33). These revisions would change the approach used by the NIH to help offset tuition, fees, and health insurance (T/F/I) costs associated with NRSA awards.
Background
The NRSA program has been used by the NIH as the primary means of supporting graduate and postdoctoral research training since enactment of the NRSA legislation in 1974. This program uses a combination of institutional training grants and individual fellowships to ensure a continuing supply of well-trained scientists prepared to conduct and lead cutting-edge health-related research.
Since its inception, the NRSA budget has grown only modestly, including during the period of doubling of the NIH budget from fiscal years (FY) 1999 to 2003. Since FY 2001, a large portion of the NRSA budget growth has gone toward higher stipends and to provide funding assistance for family health insurance to all eligible NRSA participants, as recommended by the National Academy of Sciences in its report, Addressing the Nation's Changing Needs for Biomedical and Behavioral Scientists . During this same period, the requested costs associated with the NRSA program have exceeded available funds. Specifically, the requested funds for T/F/I for T32 institutional training grants, the largest component of the NRSA program, have been increasing annually by 7% and 11%, respectively, for predoctoral and postdoctoral trainees. Barring any adjustments, continuation of this trend will result in a significant annual decrease in the number of NRSA training positions, and to fewer programs supported by T32 awards. Since these outcomes could have a substantial disruptive effect on the intended purpose of the program to build research capacity, the NIH is proposing to modify its approach to the funding of T/F/I under its NRSA programs.
Last November, the NIH held a Town Hall Meeting to hear comments and insights concerning NRSA fiscal policies. The meeting focused primarily on the funding of costs for T/F/I provided through T32 awards. Under the current formula, the NIH reimburses 100% of the requested T/F/I costs up to $3,000 and 60% of the costs above $3,000, per trainee. This formula has been employed since FY 1996 (for institutional training grants) and FY 1997 (for individual fellowships) with an adjustment in FY 2000 to partially offset additional costs of family health insurance. Several potential modifications to the NIH's current T/F/I formula were discussed at the NRSA Town Hall meeting.
The NIH has considered the feedback from the meeting participants and other stakeholders, evaluated and modeled several options, and drafted a proposed new policy (see below) for the reimbursement of costs associated with T/F/I categories.
In evaluating and modeling options for T/F/I costs, the NIH has strived to reduce or eliminate the steady decline in the overall NRSA training positions that would occur if no adjustments are made to the current policy. When computational modeling was used to assess the consequences of the proposed policy adjustments on competing NIH T32 awards made in FY 2005, it was found that an additional 135 T32 training slots could have been awarded with the funds actually expended on those awards in FY 2005. Other modeling showed the combined effect of the proposed adjustments and other factors on the yearly funding of T32 slots for the future. As the graph shows, the proposed adjustments are expected to reduce substantially (but not eliminate) the loss of future T32 full-time training positions.
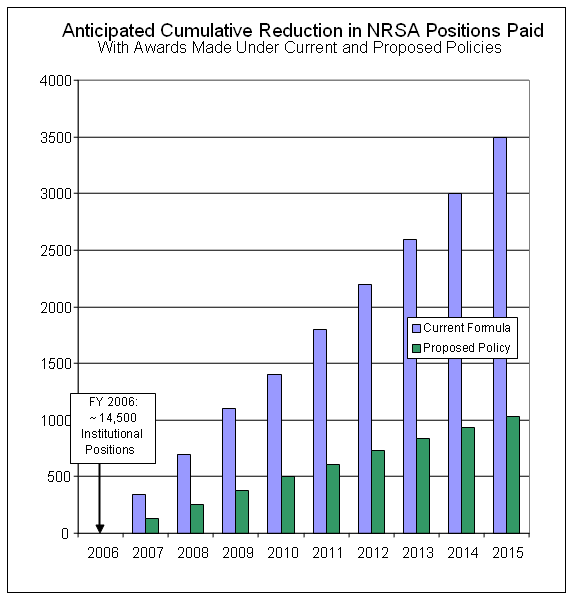
Assumptions for out-year projections shown above:
- Level NIH institutional training grant budget
- No increases in stipends provided through training grants
- Under current formula requested tuition fees and health insurance costs continue to rise at current estimated rates (7% annually for predoctoral trainees, 11% for postdocs); no increases in training related expenses.
- Under proposed policy: all future requests for tuition, fees and health insurance were assumed to rise annually by 7% (pre-doctoral) or 11% (post-doctoral), but payments would be governed by the proposed policy, which includes a new formula and caps for tuition and fees and a new category, health insurance and training
related expenses; the latter was assumed to grow in the range of 3% as the need and budget resources dictate. - Variance in competing applicants requested costs remains similar to 2005 distribution.
Prior to finalizing a new policy regarding funding of T/F/I costs, the NIH is soliciting additional comments from stakeholders. A Web site for this purpose is available at http://grants.nih.gov/grants/guide/rfi_files/NOT-OD-06-064_rfi_add.htm. Anonymous responses are permitted; however, the NIH cannot guarantee the confidentiality of the information provided. The comments provided will be analyzed and may appear in various reports.
The NIH staff will continue to assess the broader recommendations of the NRSA Town Hall meeting participants as they further evaluate additional policy options.
Proposed Policy Revisions
The NIH proposes to revise its approach to funding tuition, fees, health insurance, training related expenses (institutional NRSA), and institutional allowance (individual NRSA). These changes are described below:
- Tuition and fees: For institutional ( T32, T34, and T35 ) and individual ( F30 and F31 ) predoctoral NRSA awards, an amount per trainee equal to 60% of the level requested by the applicant institution, up to $16,000 per year, will be provided. If the program supports formally combined dual-degree training (e.g., M.D.-Ph.D, D.D.S.-Ph.D.), the amount provided per trainee will be up to $21,000 per year. For institutional ( T32, T34, and T35 ) and individual ( F32 and F33 ) postdoctoral awards, an amount per trainee equal to 60% of the level requested by the applicant institution, up to $4,500 per year, will be provided. If the program supports individuals in formal degree-granting training, the amount provided per trainee enrolled in a degree-granting program will be up to $16,000 per year.
- Training related expenses: For institutional training grants ( T32, T34, and T35 ), the training related expenses category will be modified to include health insurance as an allowable expense. An additional $2,000 per predoctoral trainee, per year, and an additional $4,000 per postdoctoral trainee, per year, will be provided in this category. This category will be referred to as health insurance and training related expenses.
- Predoctoral Institutional Allowance: For individual predoctoral fellowships ( F30 and F31), the institutional allowance category will be modified to include health insurance as an allowable expense. The NIH proposes to provide additional funding in this category so that it is comparable to that provided via the new health insurance and training related expenses category of predoctoral institutional training grants. Specifically, an additional $1,450 per predoctoral fellow per year will be provided in this category which will be referred to as health insurance and institutional allowance.
- Postdoctoral Institutional Allowance: For individual postdoctoral fellowships ( F32 and F33 ), the current institutional allowance category already includes health insurance as an allowable cost. The NIH proposes to adjust the funding provided under this category so that it is comparable to that provided via the new health insurance and training related expenses category of postdoctoral institutional training grants. Specifically, an additional $850 per postdoctoral fellow, per year, will be provided in this category which will be referred to as health insurance and institutional allowance.
Implementation Plan
The new NIH policy regarding the funding of tuition, fees, and health insurance for NRSA institutional training grants and individual fellowships will be effective with competing NRSA awards issued in FY 2007. The NIH will consider biennial adjustments to the caps and categorical funding levels for competing NRSA awards in light of needs and available resources.Responses
Comments regarding the proposed policy modifications can be provided using a special website available at http://grants.nih.gov/grants/guide/rfi_files/NOT-OD-06-064_rfi_add.htm. Responses will be accepted until Friday, June 2, 2006.
This solicitation for comments is for planning purposes only and should not be construed as a solicitation for applications or as an obligation on the part of the government. The government will not pay for the preparation of any information submitted or for the government's use of that information.
Respondents will receive an automated email notification acknowledging receipt of their response, but will not receive follow-up information on NIH's assessment of the information received. No basis for claims against the NIH shall arise as a result of a response to this solicitation for comments, or in the NIH's use of such information as either part of its evaluation process or in developing any subsequent policy or announcement.
Inquiries
Inquiries concerning this Notice may be directed to:
Office of Extramural Programs
National Institutes of Health
Email: [email protected]

